How July 3rd 2015 just changed the future of lab testing
On Friday July 3, an Arizona law allowing patients to order blood tests without involving a doctor went into effect. The law was co-authored by Theranos, a company that hopes to revolutionize the blood testing industry, worth $60 billion a year, currently dominated by LabCorp and Quest Diagnostics.
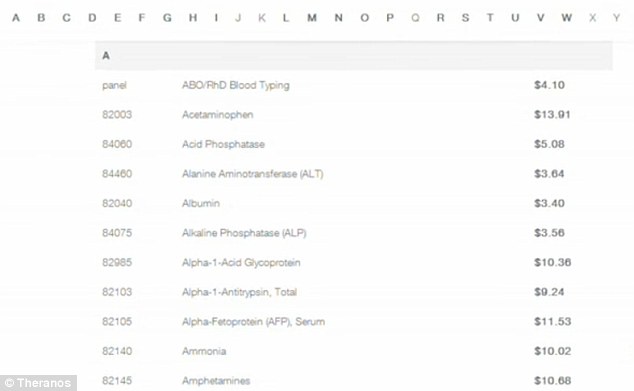
Theranos, a company based in Palo Alto, California, is the brainchild of Elizabeth Holmes, a 31 year-old Stanford University dropout considered by Forbes to be the youngest self-made billionaire in the world. The company performs blood tests using only drops of blood in tiny “Nanotainers.” Not only do the blood tests only require a finger prick instead of a blood draw, most cost less than $10 (a cholesterol panel costs only $3.40). Of note, a smaller and newer company, Healthtell of San Ramon, provides similar testing by using drops of blood on semiconductor chips coated in peptides to detect antibodies.
The technology being used and the diseases being tested for are a separate story altogether and how2med will be doing a series of articles on lab tests for disease detection and personalized medicine in the near future.
The idea of bypassing doctors is not new. In 20 states, there is already no requirement for a doctor’s order for lab tests. A number of websites and companies exist that allow patients to order their own tests (the largest being WellnessFX of San Francisco and DirectLabs of Louisiana). Some companies circumvent the requirement for a doctor by having a physician on staff to sign off on any labs a consumer wants. A few months ago, partially in response to Theranos, LabCorp announced that it will allow consumers to order their own blood tests online. In several other countries, consumers can already choose their own lab tests without doctor’s orders. So what’s new?
They have a pilot partnership with Walgreen’s pharmacies to provide their lab tests in house. Labcorps has decided not to partner with any pharmacies and Quest stopped a failing trial partnership with CVS in 2002. The difference here is that with Theranos, tests are cheap and no blood draw is needed. This makes consumers much more likely to start ordering and using these tests.
THE GOOD
Here at how2med we are excited about the opportunity to empower patients. With the Internet, big data, and mobile devices providing easy collection of data, basic interpretation of data, and cheap telemedicine with physicians to recommend and interpret lab tests, this is perfect. You can now talk to a doctor on an iPhone app, the doctor recommends a lab test, you go to Walgreens to get your finger poked, and you have the result and a recommended treatment all without involving a middle man, namely insurance companies, doctor’s offices, or appointments.
For patient’s who need monthly or weekly tests, bypassing appointments and blood draws is huge. Theranos has actually already started doing this for Pfizer and GlaxoSmithKline patients undergoing drug trials that require multiple blood tests a week.
Some doctors worry about the implications of a patient ordering a test and the patient’s primary physician then being responsible for the result of a test they never ordered. However, the bill in Arizona also protects physicians from being sued. While doctors can see their patient’s results through Theranos’s MD Connect, they are not held responsible for interpreting them. Such a law will need to be a part of the future of consumer-driven lab testing.
THE BAD
Interpretation of some tests is very complex. While home pregnancy testing and home HIV testing have proven that consumers can use and interpret tests themselves, these are simple yes/no tests. Unfortunately, most blood tests are more complex.
Tests for many diseases such as celiac disease, allergies, and even screening for cancers (such as PSA screening for prostate cancer) can often have false positives. The tests can often be positive even though the patient does not have the disease. These tests should not be ordered unless a patient actually fits certain clinical criteria, and even then the test results need to be interpreted in conjunction with other tests and exam findings. If patients without the appropriate knowledge and understanding start ordering tests, the result could be a great deal of stress and extra testing (a false positive test may necessitate a CT scan, a colonoscopy, a surgery). Finally, The tests may ultimately lead to extra costs in a healthcare system that is already straining under ballooning costs.
It will become very difficult to stop companies from marketing directly to consumers, trying to convince them that they absolutely need to have test x, y, or z.
THE FUTURE – WHAT’S NEXT?
Consumer-directed laboratory testing is likely here to stay. It is going to be an integral part of the future rapid, personalized medicine. However, its use is going to have to be very closely regulated to make certain it remains safe and effective.
For further reading:
http://www.bloomberg.com/news/
http://www.usatoday.com/story/
http://www.economist.com/news/
http://www.kevinmd.com/blog/



Your Thoughts